AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic orbitals10/26/2022  
These four quantum numbers are the principal quantum number, the azimuthal quantum number, the magnetic quantum number, and the electron spin quantum number. Thus, by determining the values of the four quantum numbers that describe an electron, one can gain insight into any electron residing in any atomic orbital in a given atom. The value of the spin quantum number, denoted by the symbol 'ms,' provides insight into the electron spin. atomic orbitals with two electrons, each electron has an equal and opposite spin to the other. 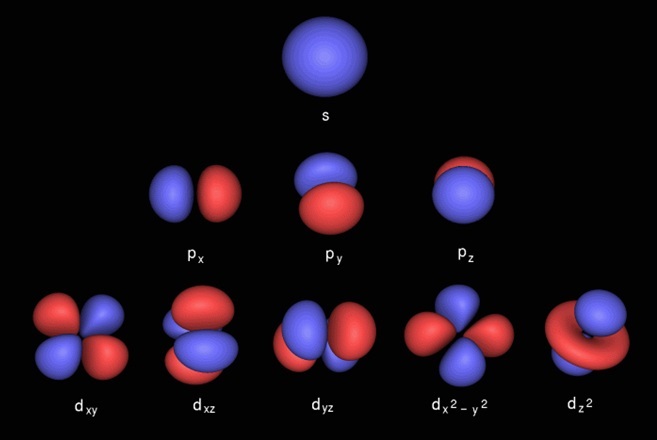
In completely occupied atomic orbitals, i.e.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
